Services
K-BPR
Active Substances
- Active Substances are chemicals, natural substances or microorganisms used to remove, detoxify or inhibit harmful organisms.
Manufacturers/importers of active substances must obtain the approval from the Minister of Environment
Notification of Existing Active Substances and Submission of Approval Application Plan
Notification of Existing Active Substances
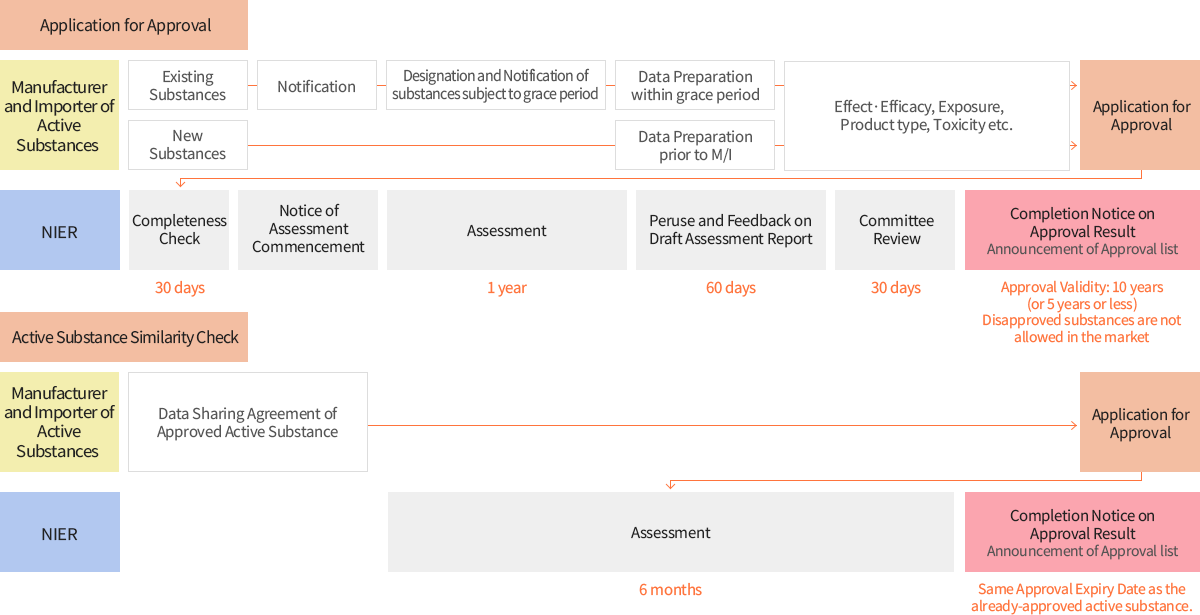
- In order to be granted an approval grace period for existing active substances distributed before December 31, 2018, manufacturers and importers shall notify to the Minister of Environment prior to manufacture/import of the active substance.
Submission of Approval Application Plan
- Submission of Approval Application Plan should be submitted within 1 year once the notified active substance is designated and announced as an existing active substance.
Notification of Existing Active Substances prior to Manufacture/Import
- Review and submission of notification
- Correspondence with NIER for supplementary
Receive Grace Period
(maximum 10 years)
Submission of Approval Application Plan
(within 1st year of notification of grace period)
- Checking for test data in Korea/overseas
- Preparation and submission of Approval Application Plan
- Correspondence with NIER for supplementary requests
Receive Approval within Grace Period
Approval of Active Substances
Joint/Individual Approval Application for Active Substances
Form CICO and Sign Agreement
- Elect Lead Registrant and form CICO
- Confirmation of eligibility for approval
- Substance sameness check
- Kick-off meeting to form CICO
- Survey on operation of CICO and approval application
- Review and execution of CICO Agreement
Data Analysis, License Purchase, Test Data Generation
- Review of available data and data owned by registrants
- Data gap analysis and key data selection
- Discussion on purchasing license to use data, and/or generating new test data
- Submission of substance approval application plan
Preparation and Submission of Application Dossiers
- Use information collection
- CICO Joint registration dossiers → Submission of Lead Registration dossiers
- Joint Registration members individual dossiers submission
* Raw materials. manufacture Process, ingredient analysis, effect/efficacy - Correspondence with NIER for supplemental data request
Post-registration Maintenance
- Sales of access to registration dossier and cost calculation for passive members joining the CICO
- Sales of access to registration dossier and cost calculation for applying substance equivalence
- Correspondence for supplemental data request
- Maintenance of change in approval such as addition of use
- Maintenance related to re-approval
| Category | CICO Joint Registration Dossier | Joint Registration Dossier by Product Types |
|---|---|---|
| Substance Information | Substance Information of active substance | - |
| Classification and Labelling | Classification and Labelling of active substance | - |
| Physico-chemical and biological properties | Required data(16) | Conditional data(4) |
| Health Hazards Environmental Hazards | Required data(24) | Conditional data(29) |
| Safety Information | Jointly prepared parts, such as substance hazard information | Product Risk and Hazard Assessment of Main Example Product |
| Handling Information | Precautions for handling and disposal method | - |
| Regulatory Information | Korean and international regulatory information | - |
Approval of Substance Equivalence
Substance Equivalence
- The technical equivalence of chemical composition, risks, and effects/efficacy of eliminating harmful organisms between different active substances
An active substance may apply for approval through substance similarity check with an already-approved active substance.
※ Same Approval Expiry Date will be applied as the already-approved active substance.
※ For Changes in Approval of the already-approved active substance, Substance Similarity Check may have to be re-approved also.
Confirmation of Availability for Substance Equivalence Check
- Existing Active Substance
 Review of Approval Criteria
Review of Approval Criteria- Application Approval for Substance Equivalence
Preparation of Data Use Agreement for Existing Active Substances
Application for Substance Equivalence Check
- Data preparation according to standards designated by MoE
Completion Notice for Substance Equivalence Check Issued

